Supported Palladium Nanoparticles that Catalyze Aminocarbonylation of Aryl Halides with Amines using Oxalic Acid as a Sustainable CO Source,Chemistry - A European Journal - X-MOL
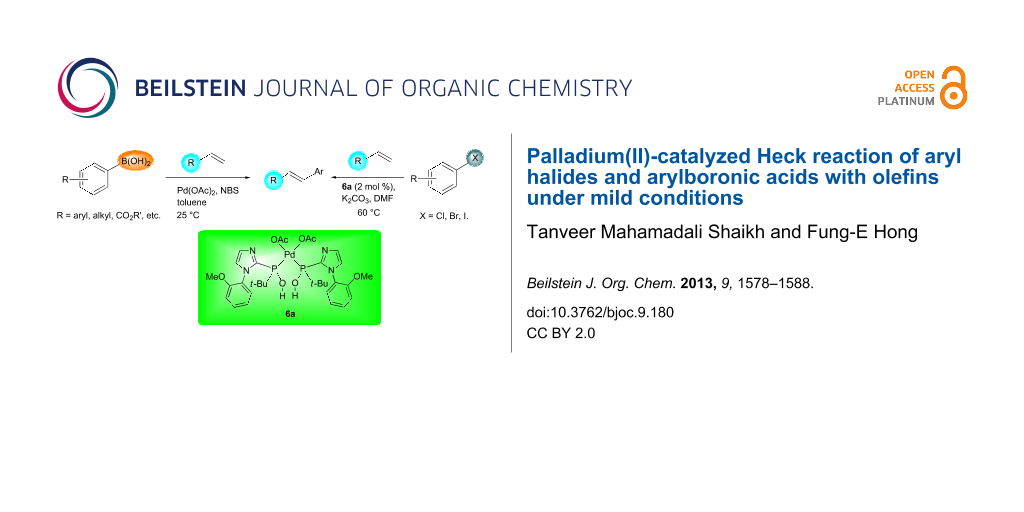
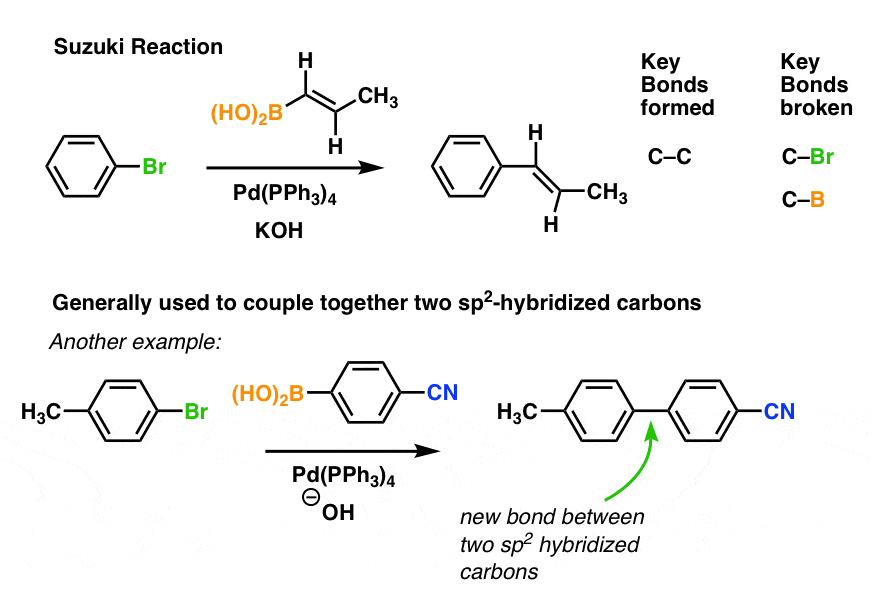
Palladium(II)-catalyzed Heck reaction of aryl halides and arylboronic acids with olefins under mild conditions

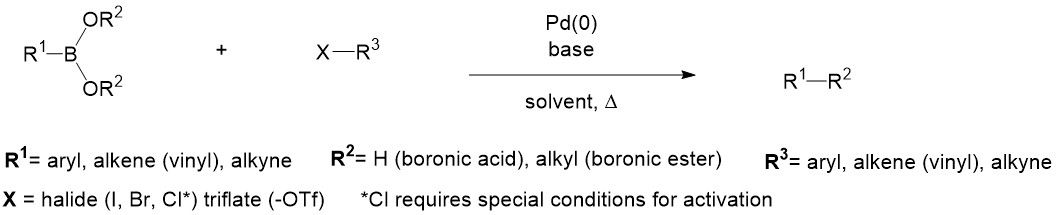
Suzuki–Miyaura Cross‐Coupling Reactions of Alkylboronic Acid Derivatives or Alkyltrifluoroborates with Aryl, Alkenyl or Alkyl Halides and Triflates - Doucet - 2008 - European Journal of Organic Chemistry - Wiley Online Library
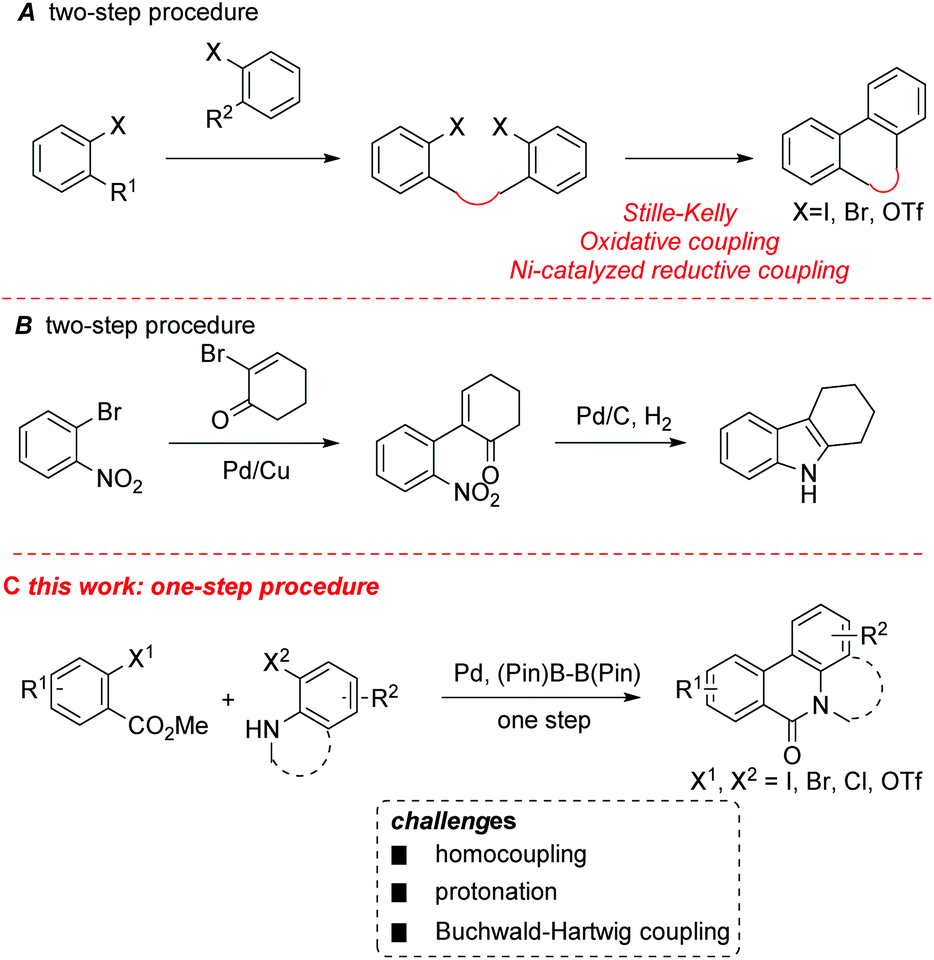
A Pd-catalyzed, boron ester-mediated, reductive cross-coupling of two aryl halides to synthesize tricyclic biaryls - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C7OB01237C

Suzuki-Miyaura cross-coupling of phenylboronic acid with aryl halides catalyzed by palladium and nickel species supported on alumina-based oxides - ScienceDirect

Studies on Pd/NiFe 2 O 4 catalyzed ligand-free Suzuki reaction in aqueous phase: synthesis of biaryls, terphenyls and polyaryls – topic of research paper in Chemical sciences. Download scholarly article PDF and

i>N</i>-Doped porous carbon supported palladium nanoparticles as a highly efficient and recyclable catalyst for the Suzuki coupling reaction

Palladium-Catalyzed, Direct Boronic Acid Synthesis from Aryl Chlorides: A Simplified Route to Diverse Boronate Ester Derivatives

Table 1 from Fibrous nano-silica (KCC-1)-supported palladium catalyst: Suzuki coupling reactions under sustainable conditions. | Semantic Scholar
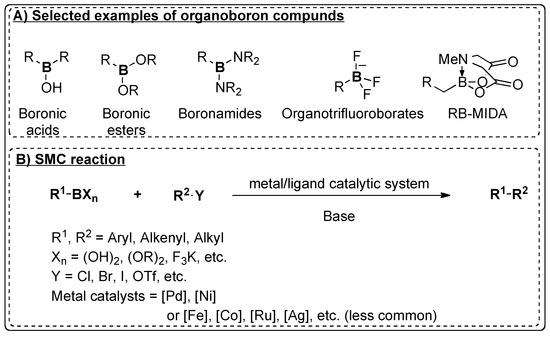
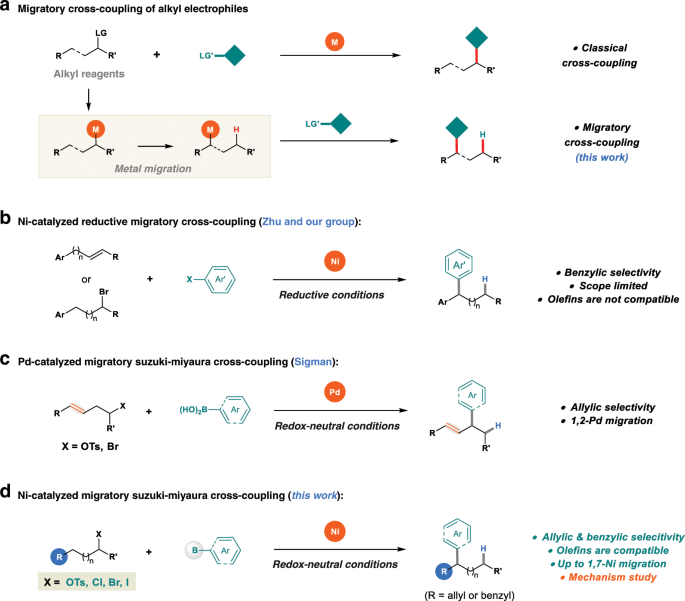
Catalysts | Free Full-Text | Recent Advances in Metal-Catalyzed Alkyl–Boron (C(sp3)–C(sp2)) Suzuki-Miyaura Cross-Couplings | HTML

Palladium‐Catalyzed Direct Arylation of (Hetero)Arenes with Aryl Boronic Acids - Yang - 2008 - Angewandte Chemie International Edition - Wiley Online Library

Pd-catalyzed Suzuki cross-Coupling reaction of bromostilbene: insights on the nature of the boron Species
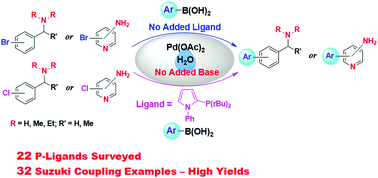
Pd-Catalyzed Suzuki coupling reactions of aryl halides containing basic nitrogen centers with arylboronic acids in water in the absence of added base - New Journal of Chemistry (RSC Publishing)

PdCl2 Immobilized in Polyacrylamide: a Low Cost and Eco-Friendly Catalyst for Suzuki-Miyaura Reactions