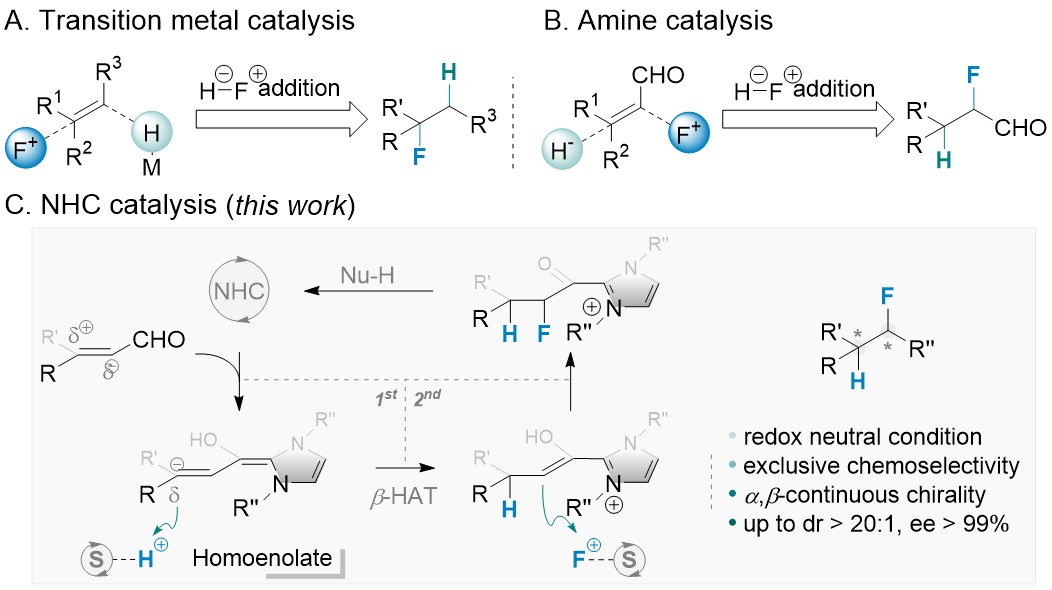
Palladium/N-heterocyclic carbene catalysed regio and diastereoselective reaction of ketones with allyl reagents via inner-sphere mechanism | Nature Communications

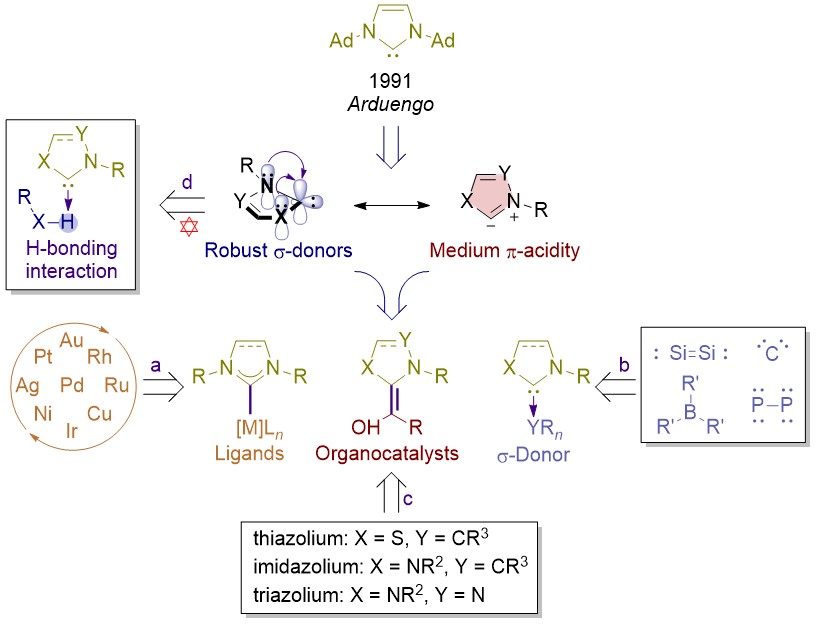
PDF) ChemInform Abstract: Sulfur-Functionalized N-Heterocyclic Carbene Complexes of Pd(II): Syntheses, Structures and Catalytic Activities

EP1022282A2 - Homo- and hetero metal alkylidene complexes of ruthenium with N-heterocyclic carbene ligands, and their use as highly active and selective catalysts for olefin metathesis - Google Patents

Palladium N-heterocyclic carbene catalyzed regioselective thiolation of 1-aryl-3-methyl-1H-pyrazol-5(4H)-ones using aryl thiols - ScienceDirect

An Antitumor Bis(N-Heterocyclic Carbene)Platinum(II) Complex That Engages Asparagine Synthetase as an Anticancer Target. - Angew. Chem. Int. Ed. - X-MOL
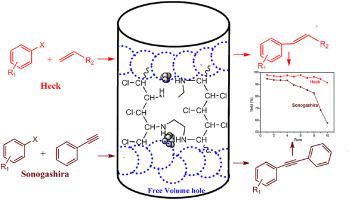
Encapsulating palladium nanoparticles inside ethylenediamine functionalized and crosslinked chlorinated poly(vinyl chloride) nanofibers as an efficient and stable heterogeneous catalyst for coupling reactions - J. Phys. Chem. Solids - X-MOL

Palladium‐Catalyzed C–S Bond Formation: Rate and Mechanism of the Coupling of Aryl or Vinyl Halides with a Thiol Derived from a Cysteine - Moreau - 2005 - European Journal of Organic Chemistry -
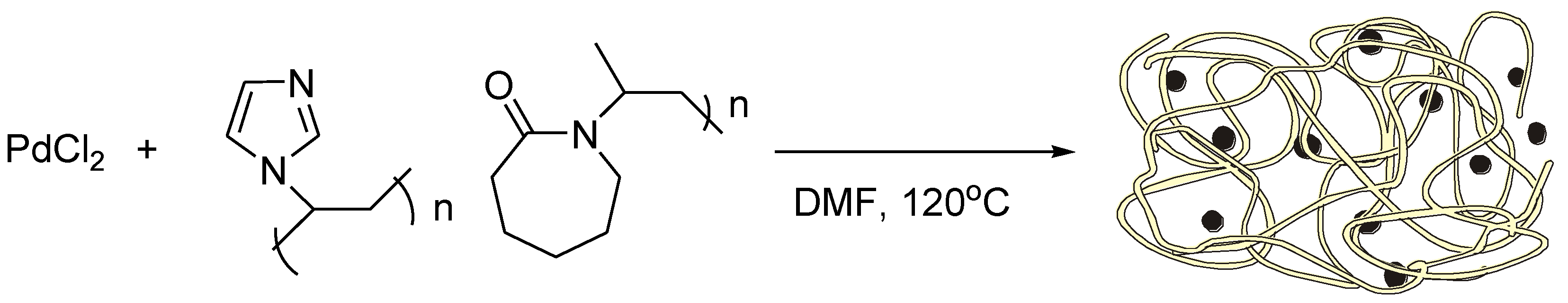
Molecules | Free Full-Text | Recyclable Nanostructured Catalytic Systems in Modern Environmentally Friendly Organic Synthesis | HTML

Palladium‐Catalyzed C–S Bond Formation: Rate and Mechanism of the Coupling of Aryl or Vinyl Halides with a Thiol Derived from a Cysteine - Moreau - 2005 - European Journal of Organic Chemistry -

Molecules | Free Full-Text | Macrocyclic Drugs and Synthetic Methodologies toward Macrocycles | HTML

Palladium‐Catalyzed C–S Bond Formation: Rate and Mechanism of the Coupling of Aryl or Vinyl Halides with a Thiol Derived from a Cysteine - Moreau - 2005 - European Journal of Organic Chemistry -

Recent advances in the catalytic applications of GO/rGO for green organic synthesis in: Green Processing and Synthesis Volume 9 Issue 1 (2020)