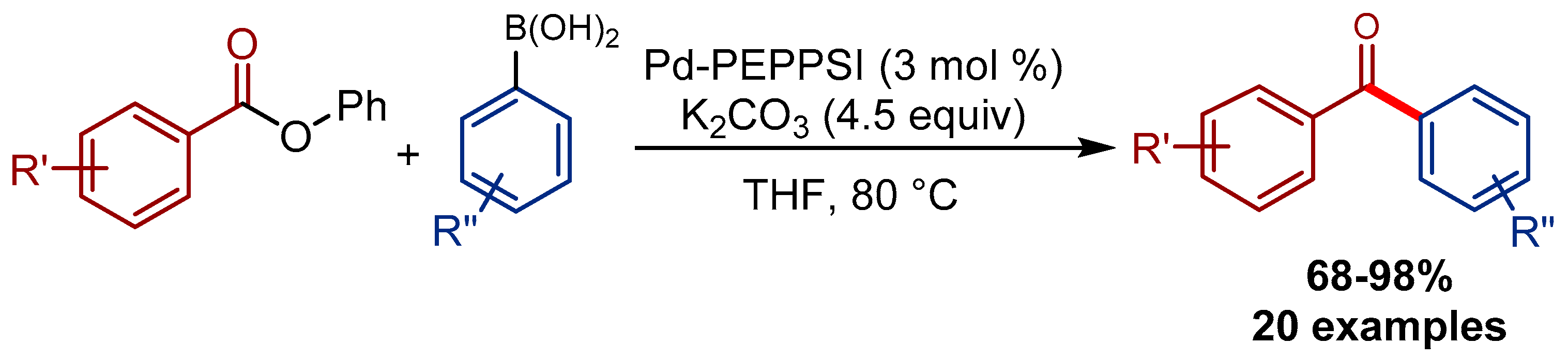Synthesis of N-heterocyclic carbene-palladium-PEPPSI complexes and their catalytic activity in the direct C-H bond activation - ScienceDirect
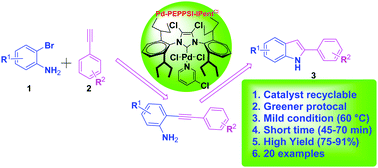
Pd-PEPPSI-IPentCl: a new highly efficient ligand-free and recyclable catalyst system for the synthesis of 2-substituted indoles via domino copper-free Sonogashira coupling/cyclization - New Journal of Chemistry (RSC Publishing)
![Catalysts | Free Full-Text | Palladium PEPPSI-IPr Complex Supported on a Calix[8]arene: A New Catalyst for Efficient Suzuki–Miyaura Coupling of Aryl Chlorides | HTML Catalysts | Free Full-Text | Palladium PEPPSI-IPr Complex Supported on a Calix[8]arene: A New Catalyst for Efficient Suzuki–Miyaura Coupling of Aryl Chlorides | HTML](https://www.mdpi.com/catalysts/catalysts-10-01081/article_deploy/html/images/catalysts-10-01081-g001.png)
Catalysts | Free Full-Text | Palladium PEPPSI-IPr Complex Supported on a Calix[8]arene: A New Catalyst for Efficient Suzuki–Miyaura Coupling of Aryl Chlorides | HTML

PDF) Phase-Separable Polyisobutylene Palladium-PEPPSI Precatalysts: Synthesis and Application in Buchwald– Hartwig Amination
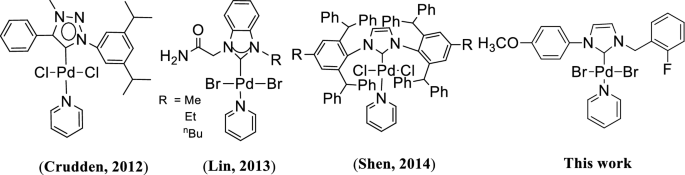
A new PEPPSI type N-heterocyclic carbene palladium(II) complex and its efficiency as a catalyst for Mizoroki-Heck cross-coupling reactions in water | SpringerLink

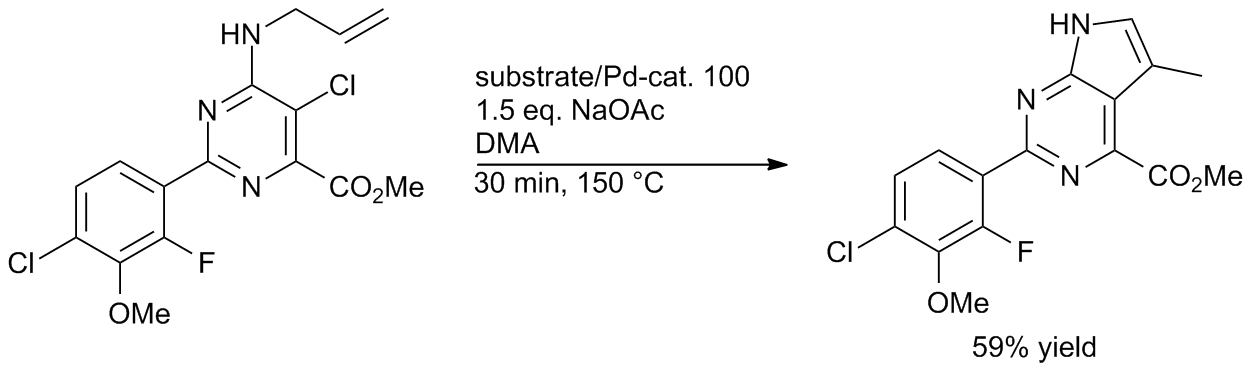
Pd‐PEPPSI: Water‐Assisted Suzuki−Miyaura Cross‐Coupling of Aryl Esters at Room Temperature using a Practical Palladium‐NHC (NHC=N‐Heterocyclic Carbene) Precatalyst - Li - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library
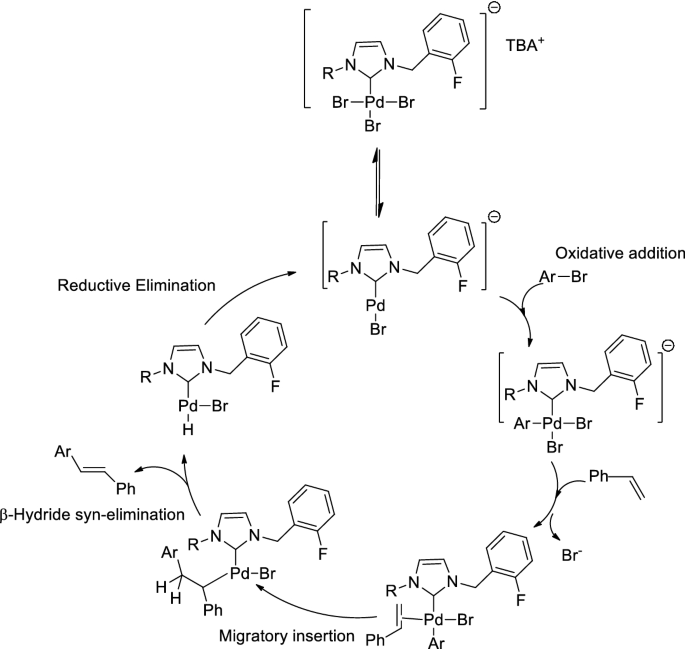
A new PEPPSI type N-heterocyclic carbene palladium(II) complex and its efficiency as a catalyst for Mizoroki-Heck cross-coupling reactions in water | SpringerLink

Synthesis, characterization and catalytic activity of PEPPSI-type palladium–NHC complexes - ScienceDirect
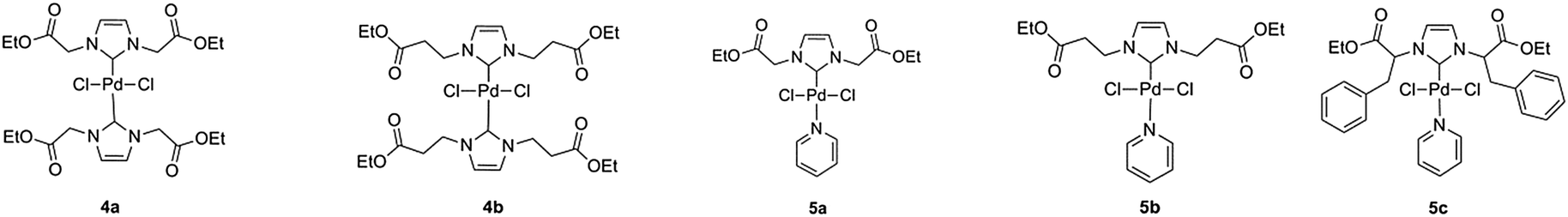
Amino acid-derived N-heterocyclic carbene palladium complexes for aqueous phase Suzuki–Miyaura couplings - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C5NJ03337C
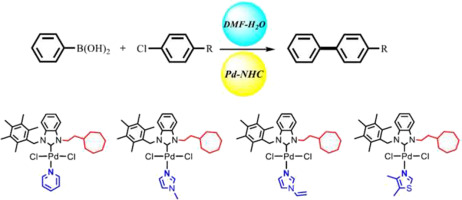
Cycloheptyl substituted N-heterocyclic carbene PEPPSI-type palladium complexes with different N-coordinated ligands: Involvement in Suzuki-Miyaura reaction - J. Organomet. Chem. - X-MOL
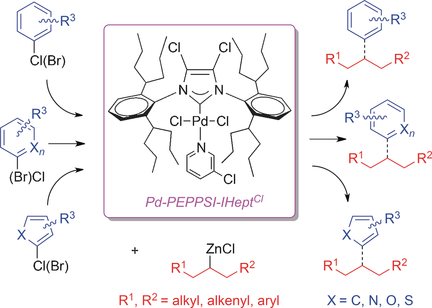
Pd‐PEPPSI‐IHeptCl: A General‐Purpose, Highly Reactive Catalyst for the Selective Coupling of Secondary Alkyl Organozincs - Chem. Eur. J. - X-MOL